| FEIS Home Page |
 |
|
| Figure 1—Evergreen blackberry in Groveland, California. Image © 2019 Barry Breckling, used with permission. |
This Species Review summarizes the scientific information about fire effects and relevant ecology of cutleaf blackberry in North America that was available as of 2020. Cutleaf blackberry occurs in western and eastern North America. It is invasive in the Pacific Northwest—particularly coastal Washington—although it is not as invasive as Himalayan blackberry. Cutleaf blackberry grows in cool temperate and semiarid climates and is both a facultative wetland and an upland species. It is most common at low elevations, on disturbed, moist to mesic sites. It grows in hardwood and conifer communities; within these communities, it is most invasive in riparian areas. It may also be invasive in riparian shrublands. Cutleaf blackberry reproduces primarily vegetatively via layering and sprouting from its rhizomes and root crown. It also reproduces from seed, which helps it establish on new sites, including burns. Its seeds are primarily dispersed by animals. The seeds have a hard coat, are dormant upon dispersal, and stored in the soil seed bank. Fire or animal ingestion helps break seed dormancy. Cutleaf blackberry is primarily an early-successional species that prefers open, disturbed sites such as streambanks, burns, clearcuts, and recently thinned or logged areas. As of 2020, fire effects on cutleaf blackberry were not documented in the literature, and there were few studies on its postfire response. It is likely that fire top-kills cutleaf blackberry, and that it sprouts after top-kill. Seeds buried in the soil seed bank are probably protected from fire. Cutleaf blackberry occurs on new burns, although its method of regeneration (from sprouts and/or seeds) is not documented. A study in the Willamette Valley of Oregon suggests that in the short term, combined density of cutleaf blackberry and Himalayan blackberry increases after one or two consecutive annual prescribed fires. The study did not distinguish between the two blackberry species. A study in Sierran mixed-conifer forests found no effect of either mastication alone, or mastication followed by prescribed fire, on basal area of cutleaf blackberry. Where cutleaf blackberry is invasive, it displaces native riparian shrubs by overtopping and outcompeting them for space, light, and nutrients. It may be controlled using a combination of treatments over many years. These may include prescribed fire, mechanical treatments, grazing, and/or herbicides. However, few studies had examined the effects of control treatments on cutleaf blackberry. |
Citation:
Fryer, Janet L. 2021. Rubus laciniatus, cutleaf blackberry.
In: Fire Effects Information System, [Online].
U.S. Department of Agriculture, Forest Service,
Rocky Mountain Research Station, Missoula Fire Sciences Laboratory (Producer).
Available: www.fs.usda.gov/database/feis/plants/shrub/rublac/all.html
[].
Taxonomy
The scientific name of cutleaf blackberry is Rubus laciniatus Willd. (Rosaceae) [49,52,66,75,78,110,116,133,150,159,164]. Cutleaf blackberry is a member of the Rubus fruiticosus complex (subgenus Rubus), an aggregate of blackberry species that are native to Eurasia [28,50] and primarily reproduce vegetatively [27]. Cutleaf blackberry is unique within the complex, and the genus, due to its deeply dissected leaflets [49] (figs. 1, 2). Within the Rubus subgenus, it is the sole member of the section Ursinus [27,69].
|
| Figure 2—Cutleaf blackberry (1, deeply divided leaflets) and Himalayan blackberry (2, oval leaflets). Forest Service, U.S. Department of Agriculture photo by Janet Fryer. |
Hybrids: Cutleaf blackberry hybridizes with Himalayan blackberry [11,28,150], Rubus bifrons, California blackberry [150], elmleaf blackberry [33,44], and woolly blackberry [44].
Common names are used throughout this Species Review, with the exception of R. bifrons. It is referred to by its scientific name to avoid confusion with the other Himalayan blackberry, R. armeniacus. See the Appendix for a complete list of plant and wildlife species mentioned in this Species Review.
SynonymsGENERAL DISTRIBUTION
Cutleaf blackberry is native to Eurasia [10,78,96,157], although its area of nativity there is uncertain [128]. It is considered nonnative in Great Britain, Scandinavia, the Mediterranean, and eastern Europe [46]. Cutleaf blackberry is cultivated for its fruits in temperate climates around the globe [10,11,94,111,133] and has established in some wildlands of North America [10,11,52,86,96], South America [96], and Australia [28,96]. It was introduced in the United States in 1860 [28,34].
Cutleaf blackberry occurs in western and eastern North America (fig. 3). In western North America, it occurs from southern coastal British Columbia [78] south to central California, southern Idaho, and northwestern Wyoming. Isolated populations occur in southern California and possibly, central Colorado [49,150], although it is rarely found in and may have disappeared from Colorado [160]. The core of cutleaf blackberry's distribution in North America is the Pacific Northwest [111,150]. It is most prevalent, and is invasive, in coastal Washington [111]. Cutleaf blackberry is absent from much of the Great Plains [111,150] and is not common in the eastern United States. It occurs sporadically from the Upper Peninsula of Michigan and upstate New York south to northern South Carolina [49,150].
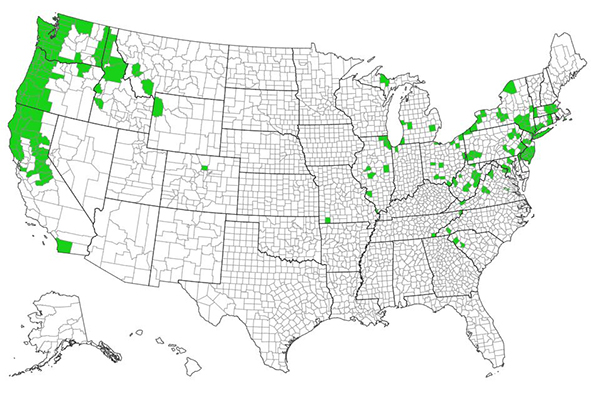 |
| Figure 3—Distribution of cutleaf blackberry in the United States. Map courtesy of EDDMaps [45] [2021, March 9]. |
Cutleaf blackberry × Himalayan blackberry and putative cutleaf blackberry × Rubus bifrons hybrids occur in coastal Oregon and Nevada County, California [11,150].
States and provinces:
United States: CA, CO, CT, DE, DC, ID, IL, IN, KY, MD, MA, MI, MO, MT, NJ, NY, NC, OH, OR, PA, RI, SC, TN, VT, VA, WA, WV, WY
Canada: BC, ON [45,74,96,150]
Cutleaf blackberry is both a facultative wetland [114,120,150] and an upland species [78,120,150]. It prefers moist to wet places such as ditches [42,78] and shores [66]. Cutleaf blackberry is flood tolerant. In British Columbia, it survived the massive, month-long 1948 flood-of-the-century on the Fraser and Columbia rivers, but it temporarily developed chlorotic leaves [16].
Cutleaf blackberry is considered an indicator of disturbed sites [78]. It grows along fencerows [57] and roadsides [11,42,78] and on rocky slopes [66]. In the Pacific Northwest, it occurs in disturbed areas [42,109,156] such as clearcuts [99] and burns [17,78,131,167]. In Michigan, it has occasionally escaped cultivation and established along roadsides, railroads, fields, and shores [116]. It is infrequent to rare in "waste" and disturbed areas in the Blue Ridge Mountains [164] and the Southeast [109,156].
Soils supporting cutleaf blackberry are typically moist to mesic [78]. Cutleaf blackberry is most common on very moist to fresh soils [11,56,78,85] that are high in humus [11] and nitrogen-rich [56,78]; it is considered an indicator species of moist, nutrient-rich soils in Vancouver, British Columbia [56]. A habitat suitability model for cutleaf blackberry on the Olympic Peninsula, Washington, predicts greater spread of cutleaf blackberry on the moist, western slope of the peninsula than on the drier eastern slope, although the authors acknowledge that spread is also possible on the eastern slope [73].
Cutleaf blackberry grows on all aspects [119] and soil textures [28,30,90], and it tolerates both acidic and alkaline soils [30]. In four riparian watersheds in western Oregon, it was not associated with topographic position (streamside, midslope/floodplain terrace, or lower hillslope) [119]. A survey in British Columbia found cutleaf blackberry grew on slopes ranging from 0° to 25°, averaging 4° [78]. Cutleaf blackberry grows in clay [28], sand, and loam [90] soils in western Oregon, and in "rocky soil" in Mt. Rainier National Park, Washington [127]. On Fire Island, New York, cutleaf blackberry is rare on dry sandflats [43].
Cutleaf blackberry grows at low elevations [42,99] (table 1). In Oregon, it is positively associated with relatively low elevations [99] and negatively associated with high elevations (R2 = 0.16, n = 252) [54]. It is more common in the Willamette Valley than in the higher-elevation Coast and Cascade ranges [5].
| Table 1—Elevational range of cutleaf blackberry in western North America. | |
| Area | Elevation (m) |
| California | <1,000 [11] |
| Colorado | reported at 1,700 [41] |
| Oregon | <1,000 [11] |
| British Columbia | 30-651; mean = 449 [78] |
Plant Communities: Cutleaf blackberry may be invasive in both hardwood and conifer communities of the Pacific Northwest, especially in riparian zones in Washington. It is not invasive in upper montane forests [7]. In the Pacific Northwest and California, it grows in riparian [118,124] and upland [22,137] hardwood communities, and in western hemlock-Sitka spruce, coastal Douglas-fir, and mixed-conifer woodlands and forests [26,37,65]. It also grows in riparian shrublands [37,70,124]; chaparral; wet [90] and mesic [66] grasslands; and on bog [118] and marsh [85] edges. Cutleaf blackberry and Himalayan blackberry often cooccur [11,68,124], although Himalayan blackberry is more prevalent and invasive [11,21,49]. Specific communities in which cutleaf blackberry occurs are described below.
Hardwood Communities: Cutleaf blackberry may be invasive in red alder communities [124], and it grows in oak woodlands and savannas [22,137] and California laurel woodlands [68]. On the Fraser River Delta, British Columbia, cutleaf blackberry and Himalayan blackberry codominate the understory of red alder riparian communities [124]. On the Dungeness and Hoh river watersheds on the Olympic Peninsula, cutleaf blackberry grows in red alder flatland communities [37]. In the Willamette Valley, it is a minor component of the low shrub strata in Oregon white oak communities [22,137]. Surveys of seven Oregon white oak communities found cutleaf blackberry was most frequent in Oregon white oak-California black oak/Pacific poison-oak forests and least frequent in Oregon white oak/Pacific poison-oak/California oatgrass savannas. Mean cover of cutleaf blackberry was ≤5% in all of the communities surveyed [22]. In the outer North Coast Ranges of southern Oregon and California, cutleaf blackberry occurs in mesic California laurel/woollyleaf manzanita woodlands [68].
Conifer Communities: Cutleaf blackberry occurs and may be invasive in western hemlock-Sitka spruce, coast Douglas-fir, and California mixed-conifer woodlands and forests [26,37,65]. On the Dungeness and Hoh river watersheds on the Olympic Peninsula, it grows in clearcuts and western hemlock-coast Douglas-fir/Pacific rhododendron forests [37]. On the Siuslaw National Forest in the Coast Ranges of Oregon, it had <2% cover in both thinned and unthinned coast Douglas-fir plantations [26].
Cutleaf blackberry grows in conifer forests east of the Coast Ranges, but it is not typically dominant or invasive. In eastern and southern Idaho, it is a minor component in Engelmann spruce/redosier dogwood and Engelmann spruce/Sitka alder/fragrant bedstraw forests [61]. It is reported as sparse in "thin woods" under the Lewis Overthrust of Glacier National Park, Montana [128], where subalpine fir-Engelmann spruce woodlands predominate [24].
Shrublands and grasslands: Cutleaf blackberry is a component of some shrublands in the Pacific Northwest and California, and it dominates some riparian shrublands in the Pacific Northwest. On the Fraser River Delta, British Columbia, cutleaf blackberry and Himalayan blackberry form thickets along river dikes [124], and cutleaf blackberry forms monoculture thickets along the Umpqua and Willamette rivers of Oregon [70]. On the Dungeness and Hoh river watersheds on the Olympic Peninsula, it grows in riparian shrublands [37], which are typically dominated by mountain alder, salmonberry, Sitka alder, and thinleaf alder [96].
Cutleaf blackberry is a component in some drier shrublands. In Mt. Tamalpais State Park, California, it grows in a coastal sage scrub community dominated by coyotebrush and the nonnative, invasive shrubs French broom and Scotch broom. Nonnative annual grasses, common periwinkle, and poison hemlock dominate the herbaceous layer. This community forms a mosaic with annual grassland [102].
Cutleaf blackberry grows on the edges of wet grasslands and on relatively dry sites within wet grasslands. In British Columbia, it grows on a bog edge with rose spirea, whitebark raspberry, and fireweed [118]. In western Oregon, it was a minor component (<4% frequency) in panicled bulrush-fowl mannagrass wetlands [90]. Near the Delaware River in Delaware, cutleaf blackberry grows in a freshwater tidal marsh [85].
See table A3 for a representative list of plant communities in which cutleaf blackberry may be invasive.Cutleaf blackberry is an evergreen shrub [78] or liana [150]. The stems (canes) arch, then droop and trail along the ground [42,78]. Flowering stems (floricanes) branch out from the main stems (primocanes) [49]. Primocanes range from 0.1 [49] to 3 m long [42,78,133]. The canes of wild-type cutleaf blackberry plants are strongly armed with numerous slightly recurved to strongly recurved prickles [52,78,110,133] (fig. 4). Some cultivars are sold as 'thornless'; however, these individuals may produce prickles after sprouting, and offspring resulting from sexual reproduction by these cultivars may also have prickles [28,58].
 |
 |
| Figure 4—Cutleaf blackberry stem. Image © 2019 Trent M. Draper, used with permission. | Figure 5—Cutleaf blackberry drupe, 3 × magnification. Image © Zoya Akulova, 1995-2021 UC Regents, used with permission. |
Cutleaf blackberry has coarse leaves [42] with prickly leaf veins and leaf stalks [42,135]. The leaves are compound, with deeply divided leaflets (figs. 1, 2). There are mostly three leaflets on primocanes [49] and mostly five leaflets on floricanes [42,109,110]. Cutleaf blackberry hybrids do not have deeply divided leaflets [162]; instead, its hybrids tend to have the oblong leaflets characteristic of other blackberries [64].
The cutleaf blackberry inflorescence is a several-flowered cyme [49,52,86]; the flowers are perfect [42,165]. The fruit is an aggregate drupe [110] of individual drupelets [11,78] (fig. 5). Each drupelet contains a single, hard-coated nutlet [165].
Stand Structure: Cutleaf blackberry is rhizomatous [143] and forms thickets [42,66,70,78,109] that may become dense. There are "almost impenetrable" cutleaf blackberry thickets along the Umpqua and Willamette rivers of Oregon [70].
Raunkiaer Life Form:Across its range in the United States, cutleaf blackberry flowers from May to August, fruits from June to September, and disperses seeds from August to November [165], depending on location (table 2).
| Table 2—Phenology of cutleaf blackberry in North America. | |
| Area | |
| United States | |
| Appalachians | flowers May-June; fruits June-July [158] |
| Blue Ridge Mountains | flowers May-June [164] |
| Northeast | flowers June-August [52,167]; fruits July-August; seeds disperse September-October [167] |
| Pacific Northwest | flowers June-August; fruits August-September; seeds disperse August-November [17] |
| Southeast | flowers May-June; fruits June-July [156] |
| California | flowers May-July [143] |
| Carolinas | flowers May-June; fruits June-July [110] |
| Delaware | flowers May-June; fruits June-July [159] |
| New York: Fire Island | flowers July [43] |
| New York: New York City, Central Park | first flowers early June [36] |
| West Virginia | flowers June-August [133] |
| Canada | |
| British Columbia | flowers early summer; fruits summer-fall [12,78] |
Reproductive Mechanisms: Blackberries, including cutleaf blackberry, have one of the most versatile systems for reproduction, colonization, and maintenance among woody plants [167]. Cutleaf blackberry reproduces vegetatively by sprouting and layering [27,100,167]. These are its primary methods of regeneration and important for population maintenance and spread. It also reproduces from seed, both sexually via pollination and asexually by apomixis. Reproduction from seed and subsequent seed dispersal are important for cutleaf blackberry's spread onto new sites [27,100].
Cutleaf blackberry × Himalayan blackberry hybrids can sprout and layer [11]. These hybrids tend to reproduce from seed sexually rather than apomictically. Their fertility is variable but is high in some individuals [11]. Cutleaf blackberry × elmleaf blackberry and cutleaf blackberry × woolly blackberry hybrids reproduce from seed both sexually and apomictically; all parents of these hybrids are nonnative [44].
Vegetative Regeneration: Cutleaf blackberry sprouts from the root crown [143] and rhizomes [88,143,161] after top-kill. It layers where stem ends touch the ground [78,143]. Most thornless cultivars of cutleaf blackberry reproduce vegetatively [28,58] but are usually sterile [28,58] and rarely reproduce from seed [28,58].
Waterways, flood waters, and landslides disperse stems of blackberries in the Rubus fruiticosus complex. The detached stems may sprout and/or layer [81,101], and establish in riparian zones after floods [108] (see Successional Status).
Pollination and Breeding System: Cutleaf blackberry is considered a facultative pseudogamous apomict because it produces seeds both sexually (via pollination) and asexually via apomixis (specifically agamospory, or formation of seeds without pollination and sexual fertilization) [11,27,28,44,58,79]. Apomixis is characteristic of blackberries native to Eurasia; blackberries native to North America do not reproduce apomictically [28].
Pollination and subsequent sexual reproduction contribute to genetic diversity in populations within the Rubus fruiticosus complex [79], including those of cutleaf blackberry. Cutleaf blackberry is dioecious [167], and it is self- and cross-pollinated [58,98,162]. A laboratory study in Sweden found viability of cutleaf blackberry pollen averaged 23% [98].
Seed Production: Age at first reproduction for cutleaf blackberry was not provided in the literature, and only one study on seed production was found. Another blackberry in the R. fruiticosus complex, R. bifrons, requires 3 years for seedlings to produce flowering canes. Mature R. bifrons plants flower in their second year [82]. A laboratory study in Sweden found cutleaf blackberry produced an average of 16 seeds/drupe. Relative seed set (percentage of ovules developing into seeds) averaged 21% [98].
Seed Dispersal: Frugivorous birds [12,167] and mammals [167] disperse cutleaf blackberry seeds. The mature fruits are rarely left unconsumed [14]. Cutleaf blackberry commonly grows along fencerows in the Pacific Northwest [57], likely due to seed dispersal by perching birds. Its establishment on the Gulf and San Juan islands of British Columbia and Washington is attributed to frugivorous birds. As of 2011, cutleaf blackberry was present on a total of 91 of these islands. The islands are small and people rarely visit them, so bird dispersal of the seeds is more likely than human dispersal. The probable dispersers include many passerine birds, but the primary dispersers are apparently American robins, European starlings, northwestern crows, song sparrows, and white-crowned sparrows [12].
Seed Banking: Cutleaf blackberry has a persistent, soil-stored seed bank [48,59,167], but it is unclear how dense its seed bank is or how long the seeds remain viable in soil. Northwestern crows store cutleaf blackberry fruits in ground-stored caches [71], and unretrieved seeds stored by crows or other seed-caching animals may form part of cutleaf blackberry's soil seed bank. In a greenhouse study using soil collected from a closed-canopy western hemlock-Sitka spruce forest on the Olympic Peninsula, cutleaf blackberry seedling emergence was low. Seedling density in the top 10 cm of soil averaged 0.07 seedling/819 cm³, and seedling frequency averaged 7.1%/819 cm³. Cutleaf blackberry seedlings did not emerge from litter samples [59].
Germination: Investigations on germination requirements of cutleaf blackberry in particular are few [38]. Blackberries are generally slow to germinate due to mechanical dormancy imposed by the hard seed coat and endocarp [38,125,167], chemical germination inhibitors in the seed coat and endocarp, and a dormant embryo [167]. Seed dormancy is broken by a combination of factors, including freeze-thaw cycles [100,167]; diurnal and annual changes in temperature (stratification) [89,167]; cycles of wetting and drying of the seed coat [167]; and scarification of the seed coat by fire [97], passage thru the digestive system of animals (i.e., acid treatment) [165,167], or damage inflicted by fungi and/or insects [167].
Field and greenhouse studies found viability of cutleaf blackberry seeds ranged from 0% to 35%, depending on treatment. Germination rates of cutleaf blackberry seeds averaged 0% after overwintering outside in boxes in a sheltered site, 14% after overwintering in a heated room, 34% after overwintering in a greenhouse, and 35% after overwintering outside in boxes at an unsheltered site. Season of planting (spring or fall) did not affect germination rates [1].
Seedling Establishment and Plant Growth: Information on seedling establishment and growth rates of cutleaf blackberry were not available as of 2020. Cutleaf blackberry's decreasing prevalence with canopy closure [78] (see Successional Status) suggests that its seedlings require open sites for establishment.
Primocanes of cutleaf blackberry gain little new length or height growth in their second year, but they develop lateral branches (floricanes) that produce flowers and fruits [167].
SUCCESSIONAL STATUS
Cutleaf blackberry is most common and may be most invasive in early succession, but it may persist into late succession [3,9,77,78,99]. In eastern and southern Idaho, its frequency was higher in early- to midseral Engelmann spruce/Sitka alder/fragrant bedstraw forests (14%) than in late-successional Engelmann spruce/redosier dogwood forests (4%) [61]. Studies along the Hoh and Dungeness rivers (Olympic Peninsula) and along Lookout Creek and the McKenzie and Willamette rivers (central Oregon) found late-successional riparian zones were less
invasible
by cutleaf blackberry and other nonnative species than riparian zones in early succession. Invasibility of riparian plant communities was estimated by the percentage of nonnative species found within the 10-year flood zone [108].
Cutleaf blackberry grows in open [66,167] to nearly closed [11,167] canopies. It is most common on open, disturbed sites [42,78,109,156] such as cutover and/or burnt sites [130,134,167], although it may also be common under partially closed canopies [11]. Colonies become more separated and infrequent as the canopy closes [78]. Cutleaf blackberry is considered shade intolerant in western hemlock-Sitka spruce forests of British Columbia. In western Oregon, cutleaf blackberry cover was negatively correlated with tree basal area (R2 = 0.9, n = 137 plots), and its frequency declined with increasing tree cover [54]. It may not be favored in gap succession. On study sites across western Oregon, it was not associated with 12- to >2,000-m2 gaps in coast Douglas-fir-Sitka spruce forests [119].
Cutleaf blackberry establishes on burned [40,78,105,130,134,167], clearcut [3,9,99], and thinned [3,107,130] sites. On the Dungeness and Hoh river watersheds on the Olympic Peninsula, it grew in clearcuts and young (<150 years old) western hemlock-coast Douglas-fir/Pacific rhododendron forests. It was not found in mature forests (>150 years old) [37]. Cutleaf blackberry establishes after logging and/or slash burning in the coastal Douglas-fir zone [130], and it is positively associated with low-elevation clearcuts [99]. On 28 sites across western Oregon, cutleaf blackberry grew in young (50-120 years), thinned Douglas-fir forests, but it was not present in young, unthinned forests or old-growth forests [9]. On the Cascade Head Experimental Forest, Oregon, cutleaf blackberry was present on a plot that had been clearcut, then planted to Sitka spruce and heavily thinned (330 trees/ha) 12 years after planting. It was not present on plots with either lighter thinning (≥648 trees/ha;) or extreme thinning (261 trees/ha) [3].
On Oregon's Coast Ranges, cutleaf blackberry had low and decreasing frequency 7 years (2%) and 10 years (1%) after a debris flow in a western hemlock-Sitka spruce forest. In contrast, native salmonberry had high and increasing frequency in postflow years 7 (69%) and 10 (74%), while Himalayan blackberry had low but increasing frequency in postflow years 7 (3%) and 10 (10%) [101].
Cutleaf blackberry occurs in old field succession [116,117]. In Olympic National Park, Washington, it was important in an old field also dominated by nonnative redtop, sweet vernalgrass, and Canada thistle. The old field was succeeding to Sitka spruce forest [117].Fire Adaptations and Plant Response to Fire: Cutleaf blackberry sprouts from the root crown [143] and rhizomes [88,143,161] after top-kill; presumably, this includes top-kill from fire. Cutleaf blackberry occurs on new burns [17,78,131,167], although its method of regeneration (from sprouts and/or seeds) is not documented. It may be common in burned areas [40,78,105,130,134,167]; fire tends to increase cutleaf blackberry cover and frequency [40,105].
Cutleaf blackberry hybrids probably also sprout after top-kill by fire. Cutleaf blackberry × Himalayan blackberry hybrids were noted in a burned riparian area in Jackson State Forest, northwestern California [11].
Cutleaf blackberry apparently requires open sites for establishment (see Successional Status). It occurs on early-seral sites such as burns [17,78,131,167], suggesting that it may establish and spread after fire. Fire can increase germination rates of blackberries in the Rubus fruiticosus complex by cracking their hard seed coats [97]; passage through an animal's digestive tract may also crack the seed coat. Because cutleaf blackberry fruits are highly palatable to and dispersed by frugivorous animals, dispersal of seeds to burns and subsequent postfire seedling establishment of cutleaf blackberry on burns is possible [3].
Studies in the Pacific Northwest and northern California show that cutleaf blackberry occurs after fire, although the studies did not provide details on its postfire abundance. It has been noted after slash burning [130] and prescribed fire [134] in the coast Douglas-fir and western hemlock-Sitka spruce zones of the Pacific Northwest. Cutleaf blackberry was present in postfire year 10 on burns in the Cascade Range in Washington and Oregon; and on the Columbia Plateau in Washington, Oregon, and Idaho [132]. In southern Oregon's Coast Ranges, it was noted in postfire years 9 and 10 after a prescribed fire on a Douglas-fir plantation [134]. In western hemlock-Sitka spruce forests of British Columbia, cutleaf blackberry was noted as "plentiful" on burnt sites, becoming more "scattered" as succession advanced [78]. In Sierran mixed-conifer forests on the Challenge Experimental Forest and in Shasta County, California, cutleaf blackberry basal area was similar on untreated control plots, masticated plots (in posttreatment year 11), and plots that had been masticated and burned under prescription (in postfire year 10) [60].
In shrubland/seasonal wetland prairie communities in the Willamette Valley, both a single fall burn and two consecutive fall burns generally increased the density of cutleaf blackberry and Himalayan blackberry in the short term. Blackberries were present on all transects where they occurred before fire; the authors did not distinguish between the two blackberry species. Compared to prefire density, mean blackberry density in postfire year 2 increased on three of four once-burned transects, and on three of four twice-burned transects (table 3). The authors speculated that in the long term, repeated burning may gradually reduce the density and slow the spread of the blackberries and other woody species that were becoming invasive in the wetland prairie [105,106].
| Table 3—Combined density of Himalayan blackberrya and cutleaf blackberrya stems on nine 3 × 30-m transects, before and after fall prescribed fire in the Willamette Valley. Once-burned sites were burned in fall 1988; twice-burned sites were burned in fall 1988 and fall 1989. Prefire and postfire data were collected in August and September of 1988 and 1990, respectively. Data are means; statistical differences were not determined. Modified from [105]. | ||||||
| Site and plant community | ||||||
| Year | 1988 | 1990 | 1988 (prefire) | 1990 | 1988 (prefire) | 1990 |
| Rose Prairie: Nootka rose/sweet vernalgrassa | 22 | 22 | 0 | 0 | 89 | 67 |
| Rose Prairie: Nootka rose/dwarf bilberry | 4 | 0 | 0 | 30 | 15 | 37 |
| Fisher Butte: Nootka rose/sweet vernalgrass | 0 | 0 | 2 | 11 | 0 | 4 |
| Fisher Butte: Nootka rose/dwarf bilberry | 7 | 7 | 6 | 33 | 2 | 9 |
| aNonnative species. | ||||||
Cutleaf blackberry leaves may provide a smaller fuel load than the leaves of native congeners. Specific leaf area (SLA) is used as a measure of leaf flammability, with low SLA associated with reduced flammability [95]. On the McDonald-Dunn Research Forest near Corvallis, mean SLA was significantly lower for cutleaf blackberry (126.65 cm²/g) than for native California blackberry and whitebark raspberry (156.21 and 221.02 cm²/g, respectively) [92,93].
Fire Regimes: The plant communities in which cutleaf blackberry occurs experience a wide variety of fire regimes. In the West, oak [146,148] and mixed-conifer communities [2,147] with cutleaf blackberry historically had a fire regime of mostly frequent, low-severity surface fires. In contrast, wet to mesic western hemlock communities historically had mostly infrequent ( ≥400-year intervals), stand-replacement fires [149]. Riparian communities had variable fire intervals and severities: fires intervals were often short and fires of low severity [145], but fires were sometimes infrequent and of mixed severity [103] or stand replacing [83].
For additional fire regime information, see FEIS publications on historical fire regimes in the following plant communities in which cutleaf blackberry may be invasive:
Hardwood Communities:For more detailed information on these topics, see the following publications: [8,18,53,151].
Because cutleaf blackberry sprouts, fire alone does not control it. Whether fire is used alone or in conjunction with other control methods, repeated treatments over many years are likely needed to control the sprouts. Cutleaf blackberry density may increase after fire ([2,105], reviews by [39,40]) but fire can be used in conjunction with other methods to increase efficacy of control treatments [2,15,125]. Agee (1986) stated that while "fire can temporarily control blackberry spread, it is not very useful in eliminating it from the site. Spot application of herbicide to remove blackberry selectively, or mowing as an alternative to burning, might be useful adjuncts to the use of fire" [2]. Repeated burning over many years may gradually reduce the density and slow the expansion of cutleaf blackberry [105,106]; however, studies of the long-term effects of fire on cutleaf blackberry were lacking as of 2020.Palatability and nutritional value: The large prickles on cutleaf blackberry stems make them unpalatable to browsing animals. In western Washington, mule deer browsed cutleaf blackberry leaves but avoided the stems [142]. Elk browse cutleaf blackberry [134], but it is not preferred. Studies in Washington and Oregon found elk avoided cutleaf blackberry browse, which comprised <1% of their diet [29]. In Redwoods State Park, California, Roosevelt elk browed cutleaf blackberry <0.4% of the time, over 4,795 minutes of forage observations [63]. However, this use was as much as expected based on availability [62]. Cutleaf blackberry leaves are palatable to domestic goats [84].
Cutleaf blackberry fruits are high in antioxidants and flavonoids such as anthocyanins and other phenolics [154]. See Ulappa (2015) for information about the nutritional content of cutleaf blackberry browse [142].
Cover value: Cutleaf blackberry thickets provide resting and hiding cover for small mammals [70] and birds, including California quail [6], brown-headed cowbirds [124], and song sparrows [124]. Many small bird species use cutleaf blackberry colonies as perching habitat [57].
Invertebrates also use cutleaf blackberry habitats. In a black willow-white ash-northern red oak riparian community in east-central Mississippi, acrobat ants nested in cutleaf blackberry thickets [140].
OTHER USES
Cutleaf blackberry is cultivated for its fruits [28,94,116]. Cultivars are commercially available [94,150], including thornless cultivars [58,94]. Blackberries are eaten fresh and made into jam, jelly, and desserts [4]. The Hoh, Quileute [113], and other tribes of the Pacific Northwest [81] have canned the fruits and eaten them raw since cutleaf blackberry established in the region [113].
Cutleaf blackberry and other blackberry species are used in ethnoveterinary medicine as a tonic and to boost lactation in dairy animals. A study at the University of Victoria found cutleaf blackberry had "midlevel" ability to boost milk production in nanny goats and cows [84].
IMPACTS
Where it is invasive, cutleaf blackberry displaces native riparian shrubs by overtopping and outcompeting them for space, light, and nutrients [91,92,115]. It may
interfere
with establishment and growth of shade-intolerant conifers [78,167] by growing over and shading out young trees. Field and laboratory experiments in Oregon found cutleaf blackberry had higher photosynthetic capacity, and maintained photosynthesis for longer in the year, than native California blackberry and whitebark raspberry [91,92].
As of 2020, cutleaf blackberry was not invasive in most of its North American distribution. It was on few state invasive species lists other than Washington (e.g., [25,31,136], see Other Status). Cutleaf blackberry is most invasive in coastal locations in Washington [111,155]; and thus, is likely to have the greatest impacts there, but it is also invasive in other parts of the Pacific Northwest. Surveys across the Cascade Range and Columbia Plateau of Washington and Oregon found cutleaf blackberry was the most common invasive woody species [55].
Cutleaf blackberry was introduced in the United States in 1860 [28,34]. Prior to the first introduction of Himalayan blackberry (Rubus armeniacus)—in 1885 in Oregon [69,76]—cutleaf blackberry was the most common nonnative blackberry in the Pacific Northwest. In the 1930s, cutleaf blackberry was still about nine times as common as Himalayan blackberry in the Pacific Northwest, but Himalayan blackberry had displaced many cutleaf blackberry and native California blackberry populations by the 1950s [28].
Cutleaf blackberry is not as invasive as Himalayan blackberry in the western United States [28,122,135]. Clark (2011) reports that cutleaf blackberry "does not have invasive status anywhere near the scale" of Himalayan blackberry [28]. Collections from Washington, Oregon, and California found Himalayan blackberry was considerably more common than cutleaf blackberry and elmleaf blackberry, with cutleaf blackberry comprising only 1% of total collections [28]. Surveys conducted from 2005 to 2009 across western Oregon found Himalayan blackberry was common and invasive but cutleaf blackberry was "not particularly common"; cutleaf blackberry was present on only 4 of 33 sites [21]. Surveyors on Myrtle Island Research Area, Oregon, reported cutleaf blackberry as "occasional" and Himalayan blackberry as "frequent" [138]. In western Oregon, FIA surveys conducted from 1995 to 1997 on nonfederal lands found cutleaf blackberry averaged 13% frequency and 5% cover, while Himalayan blackberry averaged 23% frequency and 18% cover. Cutleaf blackberry was one of eight nonnative species most commonly found on 1,127 plots, but Himalayan blackberry was the most commonly found nonnative species [54].
PREVENTION
Preventing cutleaf blackberry invasion is the most economically and ecologically effective management strategy. Maintaining the integrity of the native plant community and mitigating the factors that enhance ecosystem invasibility is likely to be more effective than solely controlling invaders such as cutleaf blackberry [67]. Minimizing soil disturbance (e.g., avoid road building in wildlands [141]), maintaining "healthy" natural communities [87,121], and monitoring several times each year [72] can help prevent its establishment, persistence, and spread. In riparian areas where reestablishment of native vegetation is the goal, a closed canopy that provides ample shade can inhibit the growth of cutleaf blackberry [13,50]. Weed prevention and control can be incorporated into many types of management plans, including those for logging and site preparation, grazing allotments, recreation management, research projects, road building and maintenance, and fire management [151]. See the
Guide to noxious weed prevention practices [151]
for specific guidelines in preventing the spread of weed seeds and propagules under different management conditions. See
Fire Management Considerations
for information on practices for preventing postfire establishment and spread of cutleaf blackberry.
CONTROL
In all cases where invasive species are targeted for control, the potential for other invasive species to fill their void must be considered no matter what method is employed [19]. Control of biotic invasions is most effective when it employs a long-term, ecosystem-wide strategy rather than a tactical approach focused on battling individual invaders [87].
Because blackberries in the Rubus fruiticosus complex—including cutleaf blackberry—sprout after top-kill, multiple entries (i.e., follow-up treatments) [35,50,126] for many years [126] are needed to control them, regardless of treatment method. In western Washington, for example, cutleaf blackberry was still present in Douglas-fir plantations that had been logged and then sprayed annually for 5 years with several different herbicides [107].
Information on controlling cutleaf blackberry was limited as of 2020. However, much of the information provided in the Species Review of Himalayan blackberry may apply to blackberries in general, including cutleaf blackberry. See the Control section in the Himalayan blackberry Species Review for more detailed information on controlling blackberries.
Fire: See the Fire Management Considerations section of this Species Review for information on preventing cutleaf blackberry establishment and spread on burned sites and on using prescribed fire to control it.
Physical or Mechanical Control: No information specific to cutleaf blackberry was available on this topic as of 2020.
Biological Control: Important considerations for developing and implementing biological control programs are provided in the Weed control methods handbook [139] and in these sources: [153,163].
Introduction of nonnative fungi and other control organisms puts native Rubus species at risk, so research in this area is not supported by the USDA [69,125]. A rust native to Europe, Phragmidium violaceum, infects cutleaf blackberry and other blackberries. However, laboratory investigations concluded that the rust does not effectively control invasive blackberries [20,21]. The rust is establishing on the West Coast. In 2005, P. violaceum was identified on Himalayan blackberries along a 160-km stretch of the Oregon Coast [129].
Chemical Control: Herbicides are effective in gaining initial control of a new invasion or a severe infestation, but they are rarely a complete or long-term solution to weed management [23,69]. Control with herbicides is temporary, because it does not change conditions that allow infestations to occur in the first place (e.g., [166]). Herbicides are most effective on large infestations when incorporated into long-term management plans that include replacement of weeds with desirable species, careful land use management, and prevention of new infestations. See the Weed control methods handbook [139] for considerations on the use of herbicides in wildlands and detailed information on specific chemicals.
MANAGEMENT UNDER A CHANGING CLIMATE| Table A1—Common and scientific names of plants mentioned in this review. Links go to other FEIS Species Reviews. | |
| Common name | Scientific name |
| western swordfern | Polystichum munitum |
| Canada thistlea | Cirsium arvense |
| fireweed | Chamerion angustifolium |
| fragrant bedstraw | Galium triflorum |
| poison hemlocka | Conium maculatum |
| California oatgrass | Danthonia californica |
| fowl mannagrass | Glyceria striata |
| panicled bulrush | Scirpus microcarpus |
| redtopa | Agrostis gigantea |
| sweet vernalgrassa | Anthoxanthum odoratum |
| common periwinklea | Vinca minor |
| Pacific poison-oak | Toxicodendron diversilobum |
| blackberries | Rubus spp., subgenus Rubus (syn. Eubatus) |
| California blackberry | Rubus ursinus |
| coyotebrush | Baccharis pilularis |
| dwarf bilberry | Vaccinium caespitosum |
| elmleaf blackberrya | Rubus ulmiforius |
| French brooma | Genista monspessulana |
| Himalayan blackberrya | Rubus armeniacus |
| Himalayan blackberrya (referred to by scientific name in the text) |
Rubus bifrons |
| mountain alder | Alnus viridis subsp. crispa |
| Nootka rose | Rosa nutkana |
| Pacific rhododendron | Rhododendron maximum |
| redosier dogwood | Cornus sericea |
| rose spirea | Spiraea douglasii |
| salmonberry | Rubus spectabilis |
| Scotch brooma | Cytisus scoparius |
| shrubby blackberry | Rubus fruticosus |
| Sitka alder | Alnus viridis subsp. sinuata |
| thinleaf alder | Alnus incana subsp. tenuifolia |
| whitebark raspberry | Rubus leucodermis |
| woolly blackberrya | Rubus tomentosus |
| woollyleaf manzanita | Arctostaphylos tomentosa |
| black willow | Salix nigra |
| California black oak | Quercus kelloggii |
| California laurel | Umbellularia californica |
| coast Douglas-fir | Pseudotsuga menziesii var. menziesii |
| Engelmann spruce | Picea engelmannii |
| Oregon ash | Fraxinus latifolia |
| Oregon white oak | Quercus garryana |
| red alder | Alnus rubra |
| Sitka spruce | Picea sitchensis |
| western hemlock | Tsuga heterophylla |
| white ash | Fraxinus americana |
| aNonnative species. | |
| Table A2—Common and scientific names of wildlife species mentioned in this review. Links go to FEIS Species Reviews. | |
| Common name | Scientific name |
| acrobat (valentine) ants | Crematogaster ashmeadi |
| Crematogaster laeviuscula | |
| American robin | Turdus migratorius |
| brown-headed cowbird | Molothrus ater |
| brown thrasher | Toxostoma rufum |
| California quail | Callipepla californica |
| European starlinga | Sturnus vulgaris |
| gallliformes | Galliformes |
| gray catbird | Dumetella carolinensis |
| gray partridge | Perdix perdix |
| northern bobwhite | Colinus virginianus |
| northern cardinal | Cardinalis cardinalis |
| northwestern crow | Corvus caurinus |
| orchard oriole | Icterus spurius |
| passerines | Passeriformes |
| pine grosbeak | Pinicola enucleator |
| ring-necked pheasanta | Phasianus colchicus |
| ruffed grouse | Bonasa umbellus |
| sharp-tailed grouse | Tympanuchus phasianellus |
| song sparrow | Melospiza melodia |
| summer tanager | Piranga rubra |
| thrushes | Turdidae |
| towhees | Emberizidae |
| yellow-breasted chat | Icteria virens |
| American black bear | Ursus americanus |
| chipmunks | Sciuridae |
| common gray fox | Urocyon cinereoargenteus |
| coyote | Canis latrans |
| elk | Cervus elaphus |
| mule deer | Odocoileus hemionus |
| northern raccoon | Procyon lotor |
| red fox | Vulpes vulpes |
| Roosevelt elk | Cervus elaphus roosevelti |
| squirrels | Sciuridae |
| skunks | Mephitidae |
| Virginia opossum | Didelphis virginiana |
| aNonnative species. | |
| Table A3—Representative plant community classifications in which cutleaf blackberry is invasive. |
| FRES20 Douglas-fir |
| FRES21 Ponderosa pine |
| FRES24 Hemlock-Sitka spruce |
| FRES28 Western hardwoods [51] |
| K001 Spruce-cedar-hemlock forest |
| K002 Cedar-hemlock-Douglas-fir forest |
| K005 Mixed conifer forest |
| K011 Western ponderosa forest |
| K012 Douglas-fir forest |
| K222 Black cottonwood-willow |
| K025 Alder-ash forest |
| K026 Oregon oakwoods |
| K028 Mosaic of K002 and K026 [80] |
| 221 Red alder |
| 222 Black cottonwood-willow |
| 223 Sitka spruce |
| 224 Western hemlock |
| 225 Western hemlock-Sitka spruce |
| 227 Western redcedar-western hemlock |
| 229 Pacific Douglas-fir |
| 230 Douglas-fir-western hemlock |
| 233 Oregon white oak |
| 234 Douglas-fir-tanoak-Pacific madrone |
| 243 Sierra Nevada mixed conifer |
| 244 Pacific ponderosa pine-Douglas-fir [47] |
| 109 Ponderosa pine shrubland |
| 203 Riparian woodland [123] |
1. Adams, John. 1927. The germination of the seeds of some plants with fleshy fruits. American Journal of Botany. 14(8): 415-428. [48174]
2. Agee, James K. 1996. Achieving conservation biology objectives with fire in the Pacific Northwest. Weed Technology. 10(2): 417-421. [94875]
3. Alaback, Paul B.; Herman, F. R. 1988. Long-term response of understory vegetation to stand density in Picea-Tsuga forests. Canadian Journal of Forest Research. 18(12): 1522-1530. [6227]
4. Alderman, DeForest C. 1979. Native edible fruits, nuts, vegetables, herbs, spices, and grasses of California: II. Small or bushy fruits. Leaflet 2278. Berkeley, CA: University of California, Division of Agricultural Sciences, Cooperative Extension. 26 p. [67652]
5. Anderson, E. William; Borman, Michael M.; Krueger, William C. 1998. The ecological provinces of Oregon: A treatise on the basic ecological geography of the state. SR 990. Corvallis, OR: Oregon Agricultural Experiment Station. 138 p. [93345]
6. Anthony, Robert. 1970. Ecology and reproduction of California quail in southeastern Washington. The Condor. 72(3): 276-287. [94686]
7. Anzinger, Dawn; Radosevich, Steven R. 2008. Fire and nonnative invasive plants in the Northwest Coastal bioregion. In: Zouhar, Kristin; Smith, Jane Kapler; Sutherland, Steve; Brooks, Matthew L., eds. Wildland fire in ecosystems: Fire and nonnative invasive plants. Gen. Tech. Rep. RMRS-GTR-42-vol. 6. Ogden, UT: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 197-224. [70906]
8. Asher, Jerry; Dewey, Steven; Olivarez, Jim; Johnson, Curt. 1998. Minimizing weed spread following wildland fires. In: Christianson, Kathy, ed. Proceedings of the Western Society of Weed Science; 1998 March 10-12; Waikoloa, HI. Volume 51. Western Society of Weed Science: 49. Abstract. [40409]
9. Bailey, John D.; Mayrsohn, Cheryl; Doescher, Paul S.; St. Pierre, Elizabeth; Tappeiner, John C. 1998. Understory vegetation in old and young Douglas-fir forests of western Oregon. Forest Ecology and Management. 112(3): 289-302. [30086]
10. Baker, William H. 1956. Plants of Iron Mountain, Rogue River Range, Oregon. The American Midland Naturalist. 56(1): 1-53. [94687]
11. Bammi, R. K.; Olmo, H. P. 1966. Cytogenetics of Rubus. v. natural hybridization between R. procerus P. J. Muell. and R. laciniatus Willd. Evolution. 20(4): 617-633. [94688]
12. Bennett, Joseph R.; Young, Emily J.; Giblin, David E.; Dunwiddie, Peter W.; Arcese, Peter. 2011. Avian dispersal of exotic shrubs in an archipelago. Ecoscience. 18(4): 369-374. [94689]
13. Bennett, Max. 2006. Managing Himalayan blackberry in western Oregon riparian areas. Corvallis, OR: Oregon State University, Oregon State University Extension Service. 16 p. [95007]
14. Bernard, Stephen R.; Brown, Kenneth F. 1977. Distribution of mammals, reptiles, and amphibians by BLM physiographic regions and A.W. Kuchler's associations for the eleven western states. Tech. Note 301. Denver, CO: U.S. Department of the Interior, Bureau of Land Management. 169 p. [434]
15. Boyce, Richard L. 2009. Invasive shrubs and forest tree regeneration. Journal of Sustainable Forestry. 28(1-2): 152-217. [74291]
16. Brink, V. C. 1954. Survival of plants under flood in the lower Fraser River valley, British Columbia. Ecology. 35(1): 94-95. [64483]
17. Brinkman, Kenneth A. 1974. Rubus L. blackberry, raspberry. In: Schopmeyer, C. S., ed. Seeds of woody plants in the United States. Agriculture Handbook No. 450. Washington, DC: U.S. Department of Agriculture, Forest Service: 738-743. [7743]
18. Brooks, Matthew L. 2008. Effects of fire suppression and postfire management activities on plant invasions. In: Zouhar, Kristin; Smith, Jane Kapler; Sutherland, Steve; Brooks, Matthew L., eds. Wildland fire in ecosystems: Fire and nonnative invasive plants. Gen. Tech. Rep. RMRS-GTR-42. Vol. 6. Ogden, UT: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: 269-280. [70909]
19. Brooks, Matthew L.; Pyke, David A. 2001. Invasive plants and fire in the deserts of North America. In: Galley, Krista E. M.; Wilson, Tyrone P., eds. Proceedings of the invasive species workshop: The role of fire in the control and spread of invasive species; Fire conference 2000: 1st national congress on fire ecology, prevention, and management; 2000 November 27 - December 1; San Diego, CA. Misc. Publ. No. 11. Tallahassee, FL: Tall Timbers Research Station: 1-14. [40491]
20. Bruckart, W. L.; Michael, J. J. 2015. Rubus armeniacus sensu stricto is not susceptible to Phragmidium violaceum in Oregon. In: Abstracts of Presentations at the 2015 Southern Division Meeting. Phytopathology. 105(4S): 21. Abstract. [94905]
21. Bruckart, William L.; Michael, Jami L.; Sochor, Michal; Travnicek, Bohumil. 2017. Invasive blackberry species in Oregon: Their identity and susceptibility to rust disease and the implications for biological control. Invasive Plant Science and Management. 10(2): 143-154. [94691]
22. Buechling, Arne; Alverson, Ed; Kertis, Jane; Fitzpatrick, Greg. 2008. Classification of oak vegetation in the Willamette Valley. Corvallis, OR: Oregon State University, Oregon Natural Heritage Information Center. 71 p. Available: http://library.state.or.us/repository/2010/201001221148005/index.pdf [2021, 3 February]. [90638]
23. Bussan, Alvin J.; Dyer, William E. 1999. Herbicides and rangeland. In: Sheley, Roger L.; Petroff, Janet K., eds. Biology and management of noxious rangeland weeds. Corvallis, OR: Oregon State University Press: 116-132. [35716]
24. Butler, David R.; Oelfke, Jack G.; Oelfke, Lori A. 1986. Historic rockfall avalanches, northeastern Glacier National Park, Montana, U.S.A. Mountain Research and Development. 6(3): 261-271. [95058]
25. California Invasive Pest Council. 2020. Cal-IPC: Plants A to Z. In: Cal-IPC, [Online]. Berkeley, CA: California Invasive Pest Council (Producer). Available: https://www.cal-ipc.org/plants/profiles/ [2020, November 22]. [94821]
26. Chan, Samuel S.; Larson, David J.; Maas-Hebner, Kathleen G.; Emmingham, William H.; Johnston, Stuart R.; Mikowski, Daniel A. 2006. Overstory and understory development in thinned and underplanted Oregon Coast Range Douglas-fir stands. Canadian Journal of Forest Research. 36(10): 2696-2711. [65481]
27. Clark, L. V.; Jasieniuk, M. 2012. Spontaneous hybrids between native and exotic Rubus in the western United States produce offspring both by apomixis and by sexual recombination. Heredity. 109(5): 320-328. [94924]
28. Clark, Lindsay Virginia. 2011. Ecological genetics of the apomictic Rubus invasion in the western United States. Davis, CA: University of California, Davis. 129 p. Dissertation. [94723]
29. Cook, John G.; Cook, Rachel C.; Davis, Ronald W.; Irwin, Larry L. 2016. Nutritional ecology of elk during summer and autumn in the Pacific Northwest. Wildlife Monographs. 195(1): 1-81. [94692]
30. Core, Earl L. 1974. Brambles. In: Gill, John D.; Healy, William M., compilers. Shrubs and vines for northeastern wildlife. Gen. Tech. Rep. NE-9. Upper Darby, PA: U.S. Department of Agriculture, Forest Service, Northeastern Forest Experiment Station: 16-19. [8923]
31. Cornell Cooperative Extension. 2014. New York state prohibited and regulated invasive plants. Ithaca, NY: Cornell University, Cornell Cooperative Extension. 13 p. [95079]
32. Crandall, Perry C. 1995. Bramble production: The management and marketing of raspberries and blackberries. Binghamton Press: The Hawthorn Press. 236 p. [95355]
33. Crane, M. B. 1940. Reproductive versatility in Rubus. I. Morphology and inheritance. Journal of Genetics. 40: 109-118. [8443]
34. Darrow, George M. 1931. European blackberry seedlings and hybrids in the Pacific Northwest. Journal of Heredity. 22(5): 143-146. [95081]
35. Davenport, Roberta. 2006. Control of knotweed and other invasive species and experiences restoring native species in the Pacific Northwest US. Native Plants Journal. 7(1): 20-26. [77567]
36. DeCandido, Robert; Calvanese, Neil; Alvarez, Regina V.; Brown, Matthew I.; Nelson, Tina M. 2007. The naturally occurring historical and extant flora of Central Park, New York City, New York 1857--2007. Journal of the Torrey Botanical Society. 134(4): 552-569. [72482]
37. DeFerrari, Collette M.; Naiman, Robert J. 1994. A multi-scale assessment of the occurrence of exotic plants on the Olympic Peninsula, Washington. Journal of Vegetation Science. 5(2): 247-258. [23698]
38. Densmore, Roseann Van Essen. 1979. Aspects of the seed ecology of woody plants of the Alaskan taiga and tundra. Durham, NC: Duke University. 285 p. Dissertation. [70495]
39. DiTomaso, Joseph M. 2006. Control of invasive plants with prescribed fire. In: DiTomaso, J. M.; Johnson, D. W., eds. The use of fire as a tool for controlling invasive plants. Cal-IPC Publication 2006-01. Berkeley, CA: California Invasive Plant Council: 7-18. [90714]
40. DiTomaso, Joseph M.; Brooks, Matthew L.; Allen, Edith B.; Minnich, Ralph; Rice, Peter M.; Kyser, Guy B. 2006. Control of invasive weeds with prescribed burning. Weed Technology. 20(2): 535-548. [63695]
41. Dittberner, Phillip L.; Olson, Michael R. 1983. The Plant Information Network (PIN) data base: Colorado, Montana, North Dakota, Utah, and Wyoming. FWS/OBS-83/86. Washington, DC: U.S. Department of the Interior, Fish and Wildlife Service. 786 p. [806]
42. Douglas, George W.; Meidinger, Del; Pojar, Jim, eds. 1999. Illustrated Flora of British Columbia: Dicotyledons (Orobanchaceae through Rubiaceae). Volume 4. Victoria, BC: Ministry of Environment, Lands and Parks and Ministry of Forests. 427 p. [94205]
43. Dowhan, Joseph J.; Rozsa, Ron. 1989. Flora of Fire Island, Suffolk County, New York. Bulletin of the Torrey Botanical Club. 116(3): 265-282. [22041]
44. Dowrick G. J. 1966. Breeding systems in tetraploid Rubus species. Genetics Research. 7(2): 245-253. [94724]
45. EDDMapS. 2020. Early detection & distribution mapping system. Athens, GA: The University of Georgia, Center for Invasive Species and Ecosystem Health. Available: http://www.eddmaps.org. [93957]
46. Euro + Med. 2021. Euro + Med PlantBase: The information resource for Euro-Mediterranean plant diversity, [Online]. Berlin, Germany: Botanic Garden and Botanical Museum, Berlin-Dahlem (Producer). Available: https://ww2.bgbm.org/EuroPlusMed/query.asp. [90645]
47. Eyre, F. H., ed. 1980. Forest cover types of the United States and Canada. Washington, DC: Society of American Foresters. 148 p. [905]
48. Ferguson, Robert B. 1983. Use of rosaceous shrubs for wildland plantings in the Intermountain West. In: Monsen, Stephen B.; Shaw, Nancy, comps. Managing Intermountain rangelands--improvement of range and wildlife habitats; Proceedings of symposia; 1981 September 15-17; Twin Falls, ID; 1982 June 22-24; Elko, NV. Gen. Tech. Rep. INT-157. Ogden, UT: U.S. Department of Agriculture, Forest Service, Intermountain Forest and Range Experiment Station: 136-149. [915]
49. Flora of North America Editorial Committee, eds. 2021. Flora of North America north of Mexico, [Online]. Flora of North America Association (Producer). Available: http://www.efloras.org/flora_page.aspx?flora_id=1. [36990]
50. Gaire, R.; Astley, C.; Upadhyaya, M. K.; Clements, D. R.; Bargen, M. 2015. The biology of Canadian weeds. 154. Himalayan blackberry. Canadian Journal of Plant Science. 95(3): 557-570. [89917]
51. Garrison, George A.; Bjugstad, Ardell J.; Duncan, Don A.; Lewis, Mont E.; Smith, Dixie R. 1977. Vegetation and environmental features of forest and range ecosystems. Agric. Handb. 475. Washington, DC: U.S. Department of Agriculture, Forest Service. 68 p. [998]
52. Gleason, Henry A.; Cronquist, Arthur. 1991. Manual of vascular plants of northeastern United States and adjacent Canada. 2nd ed. New York: New York Botanical Garden. 910 p. [20329]
53. Goodwin, Kim; Sheley, Roger; Clark, Janet. 2002. Integrated noxious weed management after wildfires. EB-160. Bozeman, MT: Montana State University, Extension Service. 46 p. Available: https://weedawareness.org/assets/documents/Integrated%20noxious%20weed%20managment%20after%20wildfires.pdf [2021, January 28]. [45303]
54. Gray, Andrew N. 2005. Eight nonnative plants in western Oregon forests: Associations with environment and management. Environmental Monitoring and Assessment. 100(1-3): 109-127. [63196]
55. Gray, Andrew. 2007. Distribution and abundance of invasive plants in Pacific Northwest forests. In: Harrington, Timothy B.; Reichard, Sarah H., tech. eds. Meeting the challenge: Invasive plants in Pacific Northwest ecosystems. Gen. Tech. Rep. PNW-GTR-694. Portland, OR: U.S. Department of Agriculture, Forest Service, Pacific Northwest Research Station: 143-150. [69636]
56. Green, R. N.; Klinka, K. 1994. A field guide to site identification and interpretation for the Vancouver Forest Region. Land Management Handbook, No. 28. Victoria, BC: Ministry of Forests Research Branch. 285 p. [87937]
57. Gullion, Gordon W. 1951. Birds of the southern Willamette Valley, Oregon. The Condor. 53(3): 129-149. [94695]
58. Hall, H. K.; Cohen, D.; Skirvin, R. M. 1986. The inheritance of thornlessness from tissue culture-derived 'Thornless Evergreen' blackberry. Euphytica. 35(3): 891-898. [6823]
59. Halpern, Charles B.; Evans, Shelley A.; Nielson, Sarah. 1999. Soil seed banks in young, closed-canopy forests of the Olympic Peninsula, Washington: Potential contributions to understory reinitiation. Canadian Journal of Botany. 77(7): 922-935. [33078]
60. Hamby, Gregory Walters. 2016. Fuels treatment longevity of mechanical mastication and growth response of ponderosa pine (Pinus ponderosa) in northern California. Starkville, MS: Mississippi State University. 65 p. Thesis. [92536]
61. Hansen, Paul L.; Hall, James B. 2002. Classification and management of USDI Bureau of Land Management's riparian and wetland sites in eastern and southern Idaho. Corvallis, MT: Bitterroot Restoration. 304 p. [82582]
62. Harper, James A. 1962. Daytime feeding habits of Roosevelt elk on Boyes Prairie, California. The Journal of Wildlife Management. 26(1): 97-100. [8876]
63. Harper, James A.; Harn, Joseph H.; Bentley, Wallace W.; Yocom, Charles F. 1967. The status and ecology of the Roosevelt elk in California. Wildlife Monographs. 16: 3-49. [83304]
64. Haskell, Gordon. 1961. Seedling morphology in applied genetics and plant breeding. Botanical Review. 27(3): 382-421. [94696]
65. Henderson, Jan A.; Peter, David H.; Lesher, Robin D.; Shaw, David C. 1989. Forested plant associations of the Olympic National Forest. R6-ECOL-TP 001-88. Portland, OR: U.S. Department of Agriculture, Forest Service, Pacific Northwest Region. 502 p. [23405]
66. Hitchcock, C. Leo; Cronquist, Arthur. 2018. Flora of the Pacific Northwest. 2nd ed. Seattle, WA: University of Washington Press. 882 p. [94186]
67. Hobbs, Richard J.; Humphries, Stella E. 1995. An integrated approach to the ecology and management of plant invasions. Conservation Biology. 9(4): 761-770. [44463]
68. Holland, Robert F. 1986. Preliminary descriptions of the terrestrial natural communities of California. Sacramento, CA: California Department of Fish and Game. 156 p. [12756]
69. Hoshovsky, Marc. 1989. Element stewardship abstract for Rubus discolor, (Rubus procerus): Himalayan blackberry, [Online]. In: Invasives on the web: The Nature Conservancy wildland invasive species program. Davis, CA: The Nature Conservancy (Producer). Available: https://www.invasive.org/gist/esadocs/documnts/rubuarm.pdf [2020, December 3]. [94947]
70. Huestis, R. R. 1932. Local differences in pelage characters of Peromyscus. Journal of Mammalogy. 13(3): 210-218. [94697]
71. James, Paul C.; Verbeek, Nicolaas A. M. 1983. The food storage behaviour of the northwestern crow. Behaviour. 85(3-4): 276-291. [94698]
72. Johnson, Douglas E. 1999. Surveying, mapping, and monitoring noxious weeds on rangelands. In: Sheley, Roger L.; Petroff, Janet K., eds. Biology and management of noxious rangeland weeds. Corvallis, OR: Oregon State University Press: 19-36. [35707]
73. Jones, Chad C.; Acker, Steven A.; Halpern, Charles B. 2007. Challenges in predicting the potential distribution of invasive species using habitat distribution models. In: Harrington, Timothy B.; Reichard, Sarah H., tech. eds. Meeting the challenge: Invasive plants in Pacific Northwest ecosystems. Gen. Tech. Rep. PNW-GTR-694. Portland, OR: U.S. Department of Agriculture, Forest Service, Pacific Northwest Research Station: 135-138. [94673]
74. Kartesz, J. T. The Biota of North America Program (BONAP). 2015. North American Plant Atlas, [Online]. Chapel Hill, NC: The Biota of North America Program (Producer). Available: http://bonap.net/napa [Maps generated from Kartesz, J. T. 2015. Floristic Synthesis of North America, Version 1.0. Biota of North America Program (BONAP). [in press]. [94573]
75. Kartesz, J. T. The Biota of North America Program (BONAP). 2015. Taxonomic Data Center, [Online]. Chapel Hill, NC: The Biota of North America Program (Producer). Available: http://bonap.net/tdc [Maps generated from Kartesz, J. T. 2010. Floristic synthesis of North America, Version 1.0. Biota of North America Program (BONAP). [in press]. [84789]
76. Kent, Douglas H. 1988. Rubus procerus 'Himalayan Giant'. Kew Magazine. 5(1): 32-36. [94951]
77. Klinka, K.; Scagel, A. M.; Courtin, P. J. 1985. Vegetation relationships among some seral ecosystems in southwestern British Columbia. Canadian Journal of Forestry. 15(3): 561-569. [5985]
78. Klinkenberg, Brian, ed. 2020. E-Flora BC: Electronic atlas of the plants of British Columbia, [Online]. Vancouver, BC: University of British Columbia, Department of Geography, Lab for Advanced Spatial Analysis (Producer). Available: https://ibis.geog.ubc.ca/biodiversity/eflora/. [54933]
79. Kollmann, Johannes; Steinger, Thomas; Roy, Barbara A. 2000. Evidence of sexuality in European Rubus (Rosaceae) species based on AFLP and allozyme analysis. American Journal of Botany. 87(11): 1592-1598. [94953]
80. Kuchler, A. W. 1964. Manual to accompany the map of potential vegetation of the conterminous United States. Special Publication No. 36. New York: American Geographical Society. 166 p. [1384]
81. Lake, Frank K. 2007. Traditional ecological knowledge to develop and maintain fire regimes in northwestern California, Klamath-Siskiyou bioregion: Management and restoration of culturally significant habitats. Corvallis, OR: Oregon State University. 732 p. Dissertation. [88360]
82. Lambrecht-McDowell, Susan C.; Radosevich, Steve R. 2005. Population demographics and trade-offs to reproduction of an invasive and noninvasive species of Rubus. Biological Invasions. 7(2): 281-295. [94954]
83. LANDFIRE Rapid Assessment. 2005. Potential Natural Vegetation Group (PNVG) R#SSHE--Sitka Spruce - Hemlock, [Online]. In: Rapid assessment reference condition models. In: LANDFIRE. Washington, DC: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station, Fire Sciences Laboratory; U.S. Geological Survey; The Nature Conservancy (Producers). Available: https://ecoshare.info/uploads/r6-analysis/reference/R_SSHE_Aug08.pdf [2021, February 8]. [83815]
84. Lans, Cheryl; Turner, Nancy; Khan, Tonya; Brauer, Gerhard; Boepple, Willi. 2007. Ethnoveterinary medicines used for ruminants in British Columbia, Canada. Journal of Ethnobiology and Ethnomedicine. 3(11): [1-22]. [77791]
85. Leck, Mary Allessio; Leck, Charles F. 2005. Vascular plants of a Delaware River tidal freshwater wetland and adjacent terrestrial areas: Seed bank and vegetation comparisons of reference and constructed marshes and annotated species list. Journal of the Torrey Botanical Society. 132(2): 323-354. [60627]
86. Lesica, Peter. 2012. Manual of Montana vascular plants. Fort Worth, TX: Brit Press. 771 p. [92949]
87. Mack, Richard N.; Simberloff, Daniel; Lonsdale, W. Mark; Evans, Harry; Clout, Michael; Bazzaz, Fakhri A. 2000. Biotic invasions: Causes, epidemiology, global consequences, and control. Ecological Applications. 10(3): 689-710. [48324]
88. Mariana, Lozinschii. 2015. Biological aspects of varieties of blackberry Chester and thornless evergreen. Conservation of Plant Diversity. 4: 28-30. [95065]
89. Markarian, D.; Olmo, H. P. 1959. Cytogenetics of Rubus. I. Reproductive behavior of R. procerus Muell. The Journal of Heredity. 50(3): 131-136. [95015]
90. McCain, Cindy; Christy, John A. 2005. Field guide to riparian plant communities in northwestern Oregon. Tech. Pap. R6-NR-ECOL-TP-01-05. [Portland, OR]: U.S. Department of Agriculture, Forest Service, Pacific Northwest Region. 357 p. [63114]
91. McDowell, Susan C. L. 2002. Photosynthetic characteristics of invasive and noninvasive species of Rubus (Rosaceae). American Journal of Botany. 89(9): 1431-1438. [42580]
92. McDowell, Susan C. L.; Turner, David P. 2002. Reproductive effort in invasive and non-invasive Rubus. Oecologia. 133(2): 102-111. [94958]
93. McDowell, Susan C. 2002. Life-history and physiological trade-offs to reproduction of invasive and noninvasive Rubus. Corvallis, OR: Oregon State University. 166 p. Dissertation. [94725]
94. McPheeters, Ken; Skirvin, Robert M. 2000. 'Everthornless' blackberry. HortScience. 35(4): 778–779. [94701]
95. Murray, Brad R.; Hardstaff, Lyndle K.; Phillips, Megan L. 2013. Differences in leaf flammability, leaf traits and flammability-trait relationships between native and exotic plant species of dry sclerophyll forest. PLoS One. 18(11): 1-8. [95023]
96. NatureServe. 2021. NatureServe Explorer, [Online]. Arlington, VA: NatureServe (Producer). Available: http://explorer.natureserve.org/. [94379]
97. Northcroft, E. G. 1927. The blackberry pest. I. Biology of the plant. New Zealand Journal of Agriculture. 34: 376-388. [95048]
98. Nybom, H. 1987. Pollen-limited seed set in pseudogamous blackberries (Rubus L. subgen. Rubus). Oecologia. 72(4): 562-568. [94702]
99. Ohmann, Janet L.; Spies, Thomas A. 1998. Regional gradient analysis and spatial pattern of woody plant communities of Oregon forests. Ecological Monographs. 68(2): 151-182. [62828]
100. Ourecky, D. K. 1975. Brambles. In: Janick, J.; Moore, J. N., eds. Advances in fruit breeding. West Lafayette, IN: Purdue University Press: 98-129. [73188]
101. Pabst, Robert J.; Spies, Thomas A. 2001. Ten years of vegetation succession on a debris-flow deposit in Oregon. Journal of the American Water Resources Association. 37(6): 1693-1708. [41709]
102. Parker, Ingrid M.; Haubensak, Karen A. 2002. Comparative pollinator limitation of two non-native shrubs: Do mutualisms influence invasions? Oecologia. 130(2): 250-258. [94712]
103. Parminter, John. 1991. Fire history and effects on vegetation in three biogeoclimatic zones of British Columbia. In: Nodvin, Stephen C.; Waldrop, Thomas A., eds. Fire and the environment: Ecological and cultural perspectives: Proceedings of an international symposium; 1990 March 20-24; Knoxville, TN. Gen. Tech. Rep. SE-69. Asheville, NC: U.S. Department of Agriculture, Forest Service, Southeastern Forest Experiment Station: 263-272. [16651]
104. Pease, Vinnie A. 1917. Duration of leaves in evergreens. American Journal of Botany. 4(3): 145-160. [94714]
105. Pendergrass, K. L.; Miller, P. M.; Kauffman, J. B. 1998. Prescribed fire and the response of woody species in Willamette Valley wetland prairies. Restoration Ecology. 6(3): 303-311. [30117]
106. Pendergrass, Kathy L. 1996. Vegetation composition and response to fire of native Willamette Valley wetland prairies. Corvallis, OR: Oregon State University. 241 p. Thesis. [66083]
107. Peter, David H.; Harrington, Constance. 2009. Six years of plant community development after clearcut harvesting in western Washington. Canadian Journal of Forest Research. 39(2): 308-319. [83132]
108. Planty-Tabacchi, Anne-Marie; Tabacchi, Eric T.; Naiman, Robert J.; Deferrari, Collette; Decamps, Henri. 1996. Invasibility of species-rich communities in riparian zones. Conservation Biology. 10(2): 598-607. [94715]
109. Pojar, Jim; MacKinnon, Andy, eds. 1994. Plants of the Pacific Northwest coast: Washington, Oregon, British Columbia and Alaska. Redmond, WA: Lone Pine Publishing. 526 p. [25159]
110. Radford, Albert E.; Ahles, Harry E.; Bell, C. Ritchie. 1968. Manual of the vascular flora of the Carolinas. Chapel Hill, NC: The University of North Carolina Press. 1183 p. [7606]
111. Randall, John. 1995. Cutleaf blackberry: Rubus laciniatus Willd., [Online]. In: IPANE: Invasive Plant Atlas of the United States. Storrs, CT: University of Connecticut, Department of Ecology and Environmental Biology (Producer). Available: https://www.invasiveplantatlas.org/subject.html?sub=6343 [2021, February 2]. [95049]
112. Raunkiaer, C. 1934. The life forms of plants and statistical plant geography. Oxford, England: Clarendon Press. 632 p. [2843]
113. Reagan, Albert B. 1934. Plants used by the Hoh and Quileute Indians. Transactions of the Kansas Academy of Science. 37: 55-70. [66487]
114. Reed, Porter B., Jr. 1988. National list of plant species that occur in wetlands: California (Region O). Biological Report 88(26.10). Washington, DC: U.S. Department of the Interior, Fish and Wildlife Service. In cooperation with: National and Regional Interagency Review Panels. 135 p. [9312]
115. Ren, M-X.; Zhang, Q-G. 2009. The relative generality of plant invasion mechanisms and predicting future invasive plants. Weed Research. 49(5): 449-460. [77029]
116. Reznicek, A. A.; Voss, E. G.; Walters, B. S. 2011. Michigan flora, [Online]. Ann Arbor, MI: University of Michigan. Available: https://michiganflora.net/home.aspx. [94671]
117. Riege, Dennis A.; del Moral, Roger. 2004. Differential tree colonization of old fields in a temperate rain forest. The American Midland Naturalist. 151(2): 251-264. [94716]
118. Rigg, George B. 1922. A bog forest. Ecology. 3(3): 207-213. [94718]
119. Sarr, D. A.; Hibbs, D. E. 2007. Woody riparian plant distributions in western Oregon, USA: Comparing landscape and local scale factors. Plant Ecology. 190(2): 291-311. [84241]
120. Schwindt, Rachel A. 2007. Plant community dynamics in remnant and restored Willamette Valley wetland prairies. Corvallis, OR: Oregon State University. 90 p. Thesis. [72287]
121. Sheley, Roger; Manoukian, Mark; Marks, Gerald. 1999. Preventing noxious weed invasion. In: Sheley, Roger L.; Petroff, Janet K., eds. Biology and management of noxious rangeland weeds. Corvallis, OR: Oregon State University Press: 69-72. [35711]
122. Sheridan, Chris D.; Spies, Thomas A. 2005. Vegetation-environment relationships in zero-order basins in coastal Oregon. Canadian Journal of Forest Research. 35(2): 340-355. [60361]
123. Shiflet, Thomas N., ed. 1994. Rangeland cover types of the United States. Denver, CO: Society for Range Management. 152 p. [23362]
124. Smith, James N. M.; Taitt, Mary J.; Zanette, Liana. 2002. Removing brown-headed cowbirds increases seasonal fecundity and population growth in song sparrows. Ecology. 83(11): 3037-3047. [94726]
125. Soll, Jonathan. 1994. Controlling Himalayan Blackberry (Rubus armeniacus [R. discolor, R. procerus]) in the Pacific Northwest. In: Ahrens, William H.; Edwards, Michael T., eds. Herbicide Handbook. 7th ed. Champaign, IL: Weed Science Society of America: 14 p. [94985]
126. Soll, Jonathan; Kreuzer, Doug; Dumont, Jason; Matthews, Ian; Hoeh, Merrit. 2008. Sandy River Riparian Habitat Protection Project report 2008. Portland, OR: The Nature Conservancy in Oregon. 50 p. [74016]
127. St. John, Harold; Warren, Fred A. 1937. The plants of Mount Rainier National Park, Washington. The American Midland Naturalist. 18(6): 952-985. [62707]
128. Standley, Paul C. 1921. Flora of Glacier National Park, Montana. Contributions from the United States National Herbarium. Vol. 22, Part 5. Washington, DC: United States National Museum, Smithsonian Institution: 235-438. [12318]
129. State of Oregon. 2021. Armenian blackberry (Himalayan blackberry). In: Oregon Noxious Weed Profiles, [Online]. Salem, OR: Oregon Department of Agriculture, Noxious Weed Control Program (Producer). Available: https://www.oregon.gov/oda/programs/weeds/oregonnoxiousweeds/pages/aboutoregonweeds.aspx#armenian-blackberry-himalayan-blackberry- [2021, March 1]. [95257]
130. Steen, Harold K. 1966. Vegetation following slash fires in one western Oregon locality. Northwest Science. 40(3): 113-120. [5671]
131. Stickney, Peter F. 1989. Seral origin of species comprising secondary plant succession in northern Rocky Mountain forests. FEIS workshop: Postfire regeneration. Unpublished draft on file at: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station, Missoula Fire Sciences Laboratory. 10 p. [20090]
132. Strand, Eva K.; Satterberg, Kevin L.; Hudak, Andrew T.; Byrne, John; Khalyani, Azad Henareh; Smith, Alistair M. S. 2019. Does burn severity affect plant community diversity and composition in mixed conifer forests of the United States Intermountain West one decade post fire? Fire Ecology. 15(25): 22 p. [94675]
133. Strausbaugh, P. D.; Core, Earl L. 1977. Flora of West Virginia. 2nd ed. Morgantown, WV: Seneca Books. 1079 p. [23213]
134. Swanson, Donald Oscar. 1970. Roosevelt elk-forest relationships in the Douglas-fir region of the southern Oregon Coast Range. Ann Arbor, MI: University of Michigan. 173 p. Dissertation. [83259]
135. Taylor, Ronald J. 1990. Northwest weeds: The ugly and beautiful villains of fields, gardens, and roadsides. Missoula, MT: Mountain Press Publishing Company. 177 p. [72431]
136. Tennessee Invasive Plant Council. 2021. TN-IPC Invasive Plant Lists, [Online]. Knoxville, TN: Tennessee Invasive Plant Council (Producer). Available: https://www.tnipc.org/invasive-plants/ [2021, February 6]. [95080]
137. Thilenius, John F. 1968. The Quercus garryana forests of the Willamette Valley, Oregon. Ecology. 49(6): 1124-1133. [8765]
138. Thompson, Ralph L. 2001. Botanical survey of Myrtle Island Research Natural Area, Oregon. Gen. Tech. Rep. PNW-GTR-507. Portland, OR: U.S. Department of Agriculture, Forest Service, Pacific Northwest Research Station. 27 p. [43785]
139. Tu, Mandy; Hurd, Callie; Randall, John M., eds. 2001. Weed control methods handbook: Tools and techniques for use in natural areas. Davis, CA: The Nature Conservancy. 194 p. [37787]
140. Tynes, James S.; Hutchins, Ross E. 1964. Studies of plant-nesting ants in east central Mississippi. The American Midland Naturalist. 72(1): 152-156. [94728]
141. Tyser, Robin W.; Worley, Christopher A. 1992. Alien flora in grasslands adjacent to road and trail corridors in Glacier National Park, Montana (U.S.A.). Conservation Biology. 6(2): 253-262. [19435]
142. Ulappa, Amy Colleen. 2015. Using foraging dynamics to answer landscape management questions: The nutritional ecology of black-tailed deer. Pullman, WA: Washington State University. 148 p. Dissertation. [94987]
143. University of California, Department of Agriculture and Natural Resources. 2020. UC-IPM: Statewide Integrated Pest Management Program. In: UC-IPM, [Online]. Davis, CA: California Invasive Pest Council (Producer). Available: http://ipm.ucanr.edu/index.html [2020, November 22]. [94822]
144. USDA, Animal and Plant Health Inspection Service. 2010. Federal noxious weed list, [Online]. In: Plant health--Noxious weeds program. Washington, DC: U.S. Department of Agriculture, Animal and Plant Health Inspection Service (Producer). Available: https://www.aphis.usda.gov/plant_health/plant_pest_info/weeds/downloads/weedlist.pdf [2021, March 23]. [36689]
145. USDA, Forest Service, Missoula Fire Sciences Laboratory. 2012. Information from LANDFIRE on fire regimes of of California oak woodlands. In: Fire Effects Information System, [Online]. U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station, Fire Sciences Laboratory (Producer). Available: https://www.fs.usda.gov/database/feis/fire_regimes/CA_oak_woodlands/all.html. [95363]
146. USDA, Forest Service, Missoula Fire Sciences Laboratory. 2012. Information from LANDFIRE on fire regimes of California coastal and valley hardwood communities. In: Fire Effects Information System, [Online]. U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station, Fire Sciences Laboratory (Producer). Available: www.fs.usda.gov/database/feis/fire_regimes/CA_coast_valley_hardwoods/all.html [2021, February 3]. [95025]
147. USDA, Forest Service, Missoula Fire Sciences Laboratory. 2018. Fire regimes of California montane mixed-conifer communities: Information from the Pacific Southwest Research Station and LANDFIRE. In: Fire Effects Information System, [Online]. U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station, Fire Sciences Laboratory (Producer). Available: www.fs.usda.gov/database/feis/fire_regimes/CA_montane_mixed_conifer/all.html. [95024]
148. USDA, Forest Service. 2012. Information from LANDFIRE on fire regimes of Oregon white oak communities. In: Fire Effects Information System, [Online]. U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station, Fire Sciences Laboratory (producer). Available: https://www.https://www.fs.usda.gov/database/feis/fire_regimes/OR_white_oak/all.html [95356]
149. USDA, Forest Service. 2012. Information from LANDFIRE on fire regimes of wet-mesic western hemlock communities. In: Fire Effects Information System, [Online]. U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station, Fire Sciences Laboratory (producer). Available: https://www.fs.usda.gov/database/feis/fire_regimes/Western_hemlock_wet/all.html [95359]
150. USDA, NRCS. 2021. The PLANTS Database, [Online]. Greensboro, NC: U.S. Department of Agriculture, Natural Resources Conservation Service, National Plant Data Team (Producer). Available: https://plants.usda.gov/. [34262]
151. USDA. 2001. Guide to noxious weed prevention practices, [Online]. Washington, DC: U.S. Department of Agriculture, Forest Service (Producer). 25 p. Available: https://www.fs.usda.gov/invasivespecies/documents/FS_WeedBMP_2001.pdf [2021, February 3]. [37889]
152. Van Dersal, William R. 1938. Native woody plants of the United States, their erosion-control and wildlife values. Misc. Publ. No. 303. Washington, DC: U.S. Department of Agriculture. 362 p. [4240]
153. Van Driesche, Roy; Lyon, Suzanne; Blossey, Bernd; Hoddle, Mark; Reardon, Richard, tech. coords. 2002. Biological control of invasive plants in the eastern United States. Publication FHTET-2002-04. Morgantown, WV: U.S. Department of Agriculture, Forest Service, Forest Health Technology Enterprise Team. 413 p. Available online: https://www.fs.usda.gov/foresthealth/technology/pdfs/BiocontrolsOfInvasivePlants02_04.pdf [2021, February 19]. [54194]
154. Wada, Leslie; Ou, Boxin. 2002. Antioxidant activity and phenolic content of Oregon caneberries. Journal of Agricultural and Food Chemistry. 50(12): 3495-3500. [94729]
155. Washington State, Noxious Weed Control Board. 2020. Noxious Weed List, [Online]. Olympia, Washington: Noxious Weed Control Board (Producer). Available: https://www.nwcb.wa.gov/printable-noxious-weed-list [2020, November 6]. [94765]
156. Weakley, Alan S. 2008. Flora of the Carolinas, Virginia, Georgia, northern Florida, and surrounding areas: Working draft of 7 April 2008. Chapel Hill, NC: University of North Carolina Herbarium (NCU), North Carolina Botanical Garden, University of North Carolina at Chapel Hill. 1015 p. Available: http://www.herbarium.unc.edu/WeakleysFlora.pdf [2011, January 19]. [81727]
157. Weakley, Alan S. 2020. Flora of the southeastern United States [Online]. Chapel Hill, NC: University of North Carolina (Producer). Available: http://herbarium.bio.unc.edu/FSUS_2020/FSUS.pdf. [94895]
158. Weakley, Alan S. 2020. Flora of the southeastern United States: Appalachian Region, [Online]. Chapel Hill, NC: University of North Carolina (Producer). Available: http://herbarium.bio.unc.edu/FSUS_2020/FSUS_Appalachian_Region.pdf. [94896]
159. Weakley, Alan S. 2020. Flora of the southeastern United States: Delaware, [Online]. Chapel Hill, NC: University of North Carolina (Producer). Available: http://herbarium.bio.unc.edu/FSUS_2020/FSUS_Delaware.pdf. [94923]
160. Weber, William A.; Whittman, Ronald C. 2012. Colorado flora: Eastern slope. A field guide to the vascular plants, 4th Edition. Boulder, CO: University Press of Colorado. 555 p. [94319]
161. Weedbusters. 2021. Weed list, [Online]. New Zealand Department of Conservation (Producer). Available: https://www.weedbusters.org.nz/. [95064]
162. Werlemark, Gun; Nybom, Hilde. 2003. Pollen donor impact on progenies of pseudogamous blackberries (Rubus subgen. Rubus). Euphytica. 133(1): 71–80. [94730]
163. Wilson, Linda M.; McCaffrey, Joseph P. 1999. Biological control of noxious rangeland weeds. In: Sheley, Roger L.; Petroff, Janet K., eds. Biology and management of noxious rangeland weeds. Corvallis, OR: Oregon State University Press: 97-115. [35715]
164. Wofford, B. Eugene. 1989. Guide to the vascular plants of the Blue Ridge. Athens, GA: The University of Georgia Press. 384 p. [12908]
165. Young, James A.; Young, Cheryl G. 1992. Seeds of woody plants in North America. [Revised and enlarged edition]. Portland, OR: Dioscorides Press. 407 p. [72640]
166. Youtie, Berta; Soll, Jonathan. 1990. Diffuse knapweed control on the Tom McCall Preserve and Mayer State Park. Grant proposal prepared for the Mazama Research Committee, Portland OR. Unpublished paper on file at: U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station, Missoula Fire Sciences Laboratory. 18 p. [38353]
167. Zasada, John C.; Tappeiner, John C., III. 2008. Rubus spp.: Blackberry raspberry. In: Bonner, Franklin T.; Karrfalt, Robert P., eds. Woody plant seed manual. Agric. Handbook No. 727. Washington, DC: U.S. Department of Agriculture, Forest Service: 984-996. [73150]